If you’re feeling depressed or anxious, usually, the last thing you tend to think about is what you’ve been eating. However, new research suggests that you’d be wise to do just that.
We used to think that the brain controlled everything. The brain gave the orders, and everything else in the body followed them. It turns out that it’s not that simple. The digestive system — or gut, as it’s also called — is lined with nerves that continuously communicate with the brain, not only in response to orders but often to give them as well.
Scientists now understand that what goes on in the gut can influence the brain to the point of affecting our emotions. If you’re feeling down today, it could have just as much to do with the meals you’ve been eating during the past week as what’s been going on in your life.
This is good news because it means that we may have another way to treat anxiety and depression, which is through dietary changes.
What Is the Gut-brain Connection?
You already know that your brain has a direct effect on your stomach. Your brain tells you you’re hungry, and your stomach growls. You feel stressed out, and your stomach churns. If you’re excited, you may feel butterflies.
You may not have realized that the communication between these two organs goes both ways. That’s because embedded in the walls of the intestines are millions of neurons responsible for sensing information and then communicating that information as needed.
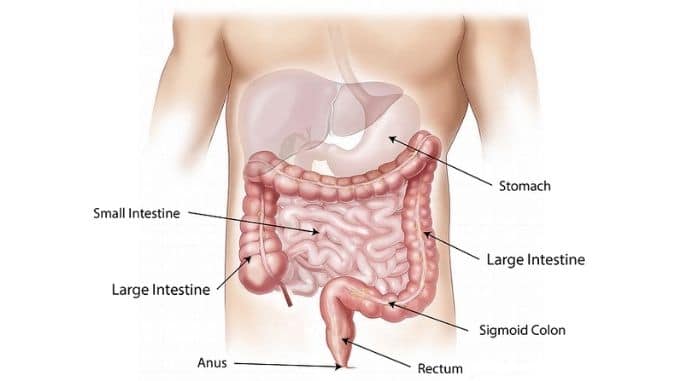
Called the “enteric nervous system” (ENS), this multitude of neurons begins in the esophagus and extends all the way through the digestive system to the end of the colon. It controls the daily task of digestion, processing food, absorbing nutrients, and expelling waste, all while managing muscle movements that keep everything moving.
Recently, however, scientists have come to the conclusion that this system does far more than simply handle the digestive process. Their instincts were proven correct when they discovered that about 90 percent of the fibers in the vagus nerve — the longest and most complex of nerves that go from the brain to the rest of the body — take information from the gut to the brain, not the other way around.
In other words, the gut and the brain are in constant communication along this vagus nerve in a system that scientists have labeled the “gut-brain axis” (GBA). It’s like a busy communication highway keeping these two in close contact about everything from digestion to the immune system to the individual’s stress and emotions.
Scientists now call the gut (or the ENS) the “second brain” because it can perform independently of the brain’s oversight. “Once dismissed as a simple collection of relay ganglia,” researchers wrote in a 1999 study, “the enteric nervous system is now recognized as a complex, integrative brain in its own right.”
So just what does this second brain do?
How the Second Brain Influences Health
Inside the gut is a complex community of bacteria. Scientists call it the “microbiome” and have discovered that the balance of good to bad bacteria that live in this community can influence not only digestion and other facets of physical health but elements of mental and emotional health too.
Usually, when we think of bacteria, we think of the bad ones that make us sick. However, there are good ones, too, that we need in the gut to feel our best. It’s hard to imagine, but scientists say that the total number of bacteria in the gut is in the trillions, with as many as 1,000 different bacterial species represented.
We pick up these microbes from our mothers when we are born and then continue to gain more via breast milk and our early diet. Around toddler age, the microbiome stabilizes but is vulnerable to things like the environment, diet, stress, illnesses, and drugs like antibiotics, all of which can affect the balance of microbes even after we become adults.
In truth, we live in a relationship with these microbes every second of our lives. If the microbiome is healthy and balanced, then we are, too. But if it becomes unbalanced, with too many “bad” bacteria and not enough “good” ones, we can suffer from a number of problems, including the following:
- Autoimmune diseases: Rheumatoid arthritis, muscular dystrophy, multiple sclerosis, type 1 diabetes, and fibromyalgia are all associated with a dysfunction in the microbiome
- Weight: Studies have found a difference between the microbiomes of obese and lean twins. Obese twins had a lower diversity of bacteria than the lean ones did; there’s also evidence that some gut microbes could influence appetite
- Inflammatory bowel disease: Diseases like ulcerative colitis and Crohn’s disease have been linked to shifts in bacterial gut populations
- Irritable bowel syndrome (IBS): Dietary changes that help influence the balance in the microbiome have been found to be successful in treating IBS in some patients
- Cardiovascular disease: There is growing evidence that there may be a link between the microbiome and cardiovascular disease
- Antibiotic-resistant infections: A Clostridium difficile (CDI) infection can be life-threatening because most of today’s antibiotics don’t work against it; it is a “superbug” that scientists have linked to an overuse of antibiotics in the general population; studies have shown that transplanting fecal matter (with healthy microbes in it) from a healthy person to a person inflicted with CDI can help cure the disease
Beyond all these effects — and potentially others — scientists have also learned that the microbiome can influence how we feel and behave, particularly when it comes to anxiety and depression.
The Link Between the Gut, Depression & Anxiety
We used to believe that depression was the result of an imbalance of certain neurotransmitters in the brain, like serotonin. More recent studies, however, have cast doubt on that theory, so scientists are looking for other explanations.
So far, evidence suggests that problems in the gut can contribute to anxiety and depression in a number of ways. If the microbiome becomes unbalanced, for example, it can affect mood.
In a 2018 study, researchers reported that the gut microbiome “has been strongly associated with mood-relating behaviors, including major depressive disorder” (MDD). They added that the link between the two was characterized by the communication between the gut and the brain, suggesting that imbalances in the microbiome could contribute to depression and that depression could also change the microbiome.
Studies have also found that inflammation in the gut is linked with mental illnesses, including anxiety and depression.
When you cut yourself, your immune cells go to work repairing the damage. This creates inflammation — that swelling, a red effect that you see. This is a good thing as long as the wound is repaired and the inflammation goes away fairly quickly.
If the problem causing the inflammation isn’t fixed, however, the inflammation may continue. Imagine a cut that just doesn’t heal. The immune system continues to work on it to no avail. Inflammation becomes “chronic” or ongoing, which has negative consequences.
Scientists now think that depression may be a symptom of chronic inflammation in the gut. If the microbiome becomes unbalanced or stressed, harmful gut microbes multiply beyond healthy levels and release toxins that trigger the immune system, leading to chronic inflammation.
In time, that inflammation can damage the intestinal wall, which then allows food particles and “bad” bacteria to leak into the bloodstream. This is called “leaky gut syndrome.” If it continues, the bad bacteria can make their way into the ENS, triggering the immune system to release more inflammatory cells to battle these “invaders.”
These inflammatory cells activate the vagus nerve and can negatively impact the activity of the brain and central nervous system. This is the type of effect that scientists believe can lead to depression and anxiety in some people.
Studies have shown, for example, that major depressive disorder is associated with persistent low-grade inflammation and with increased levels of circulating inflammatory cells. Studies have also shown depressed patients to have altered microbiomes containing more inflammatory populations of bacteria.
There are a number of different factors involved that affect one another, and it can all get quite complicated. What matters most is that we understand that the health of the gut and the microbiome, in particular, can impact mental health.
7 Gut-focused Ways to Prevent and Treat Anxiety and Depression
It makes sense, then, that if we can improve gut health, we might be able to improve mental health too. Scientists are beginning to find new ways to treat depression based on repairing the microbiome and the intestinal wall.
In a 2017 review of 10 studies, researchers found that treatment with probiotics — which help improve the balance of good-to-bad bacteria in the gut — helped alleviate symptoms of depression. Researchers warned that more studies are needed but suggested the therapy was promising. Probiotics have also been found to help reduce inflammation and to help normalize the action of the immune system.
The following seven steps are also believed to help maintain and improve gut health, which may, in turn, help improve other facets of your health as well.
1. Take Probiotics
These may help restore an unbalanced microbiome. Eat more foods rich in probiotics like sauerkraut, yogurt, kefir, miso, pickles, tempeh, kimchi, and kombucha. If you’re already experiencing depression, talk to your doctor about probiotic supplements.
2. Include Omega-3 Fatty Acids in Your Diet
These are known anti-inflammatories and may also help maintain the health of the intestinal wall, reducing leaky gut. Eat more fatty fish, walnuts, flaxseeds, oysters, and chia seeds.
3. Get Enough Fiber
It helps keep the digestive system healthy and feeds the good bacteria.
4. Sleep Is Essential
While you’re sleeping, your body repairs any problems that may exist. Without enough sleep, inflammation increases, which can make gut imbalances worse.
5. Reduce Intake of Inflammatory Foods
Fast foods, fried foods, and high-fat and high-sugar foods are all linked with increased inflammation. Decrease intake of these and focus on eating healthy, whole foods.
6. Consider Anti-inflammatory Supplements
Always check with your doctor first, but then if you feel you may be battling inflammation, try supplements like turmeric, L-glutamine, and licorice root to help reduce inflammation and heal the gut.
7. Manage Your Stress
Chronic stress has been linked with inflammation and the release of hormones that can damage the microbiome and the intestinal wall. Practice stress-relieving activities like yoga, meditation, tai chi, long walks, journaling, deep breathing, pet therapy, and time with friends and loved ones.
For your guide to the best foods to heal your body, check out The Best Foods That Rapidly Slim & Heal in 7 Days here!